Eye development
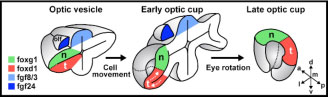
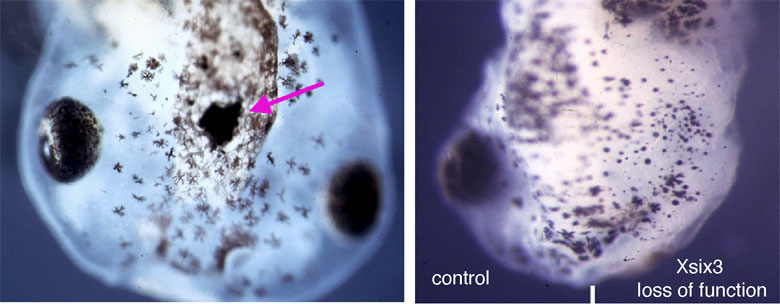
The eyes enable organisms to visually perceive their surroundings. During embryogenesis, the eyes originate as outpocketings of the brain and our work aims to elucidate the key stages of eye development. These include induction of the eye field, outgrowth and morphogenesis of the optic vesicles, formation and closure of the choroid fissure, growth of the eye and differentiation of retinal neurons. In addition to deepening our understanding of normal eye development, our studies are addressing how this process can go wrong and lead to congenital eye defects. In particular, we are studying the genetic and cellular bases of MAC phenotypes (microphthalmia, anophthalmia and coloboma). These studies are helping to understand the aetiology of eye diseases and hereditary visual system conditions in human patients.
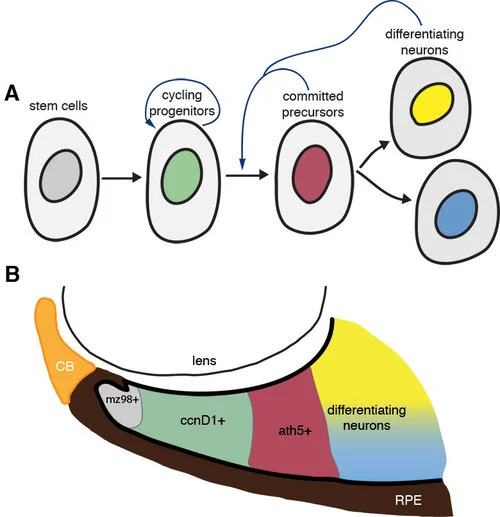
Complementing our studies of eye formation and differentiation we are also studying cell behaviour in a retinal stem cell niche. In the post-embryonic zebrafish eye, all the stages of progression from stem cell to differentiated neuron are found near the margin of the eye, in a region termed the ciliary marginal zone (CMZ). We are interested in exploring the molecular mechanisms that govern neurogenesis in the CMZ and how specific intrinsic and extrinsic signals are integrated to carry out this task and allow the growth of the eye.
RELATED PUBLICATION SUMMARIES
EYE DEVELOPMENT PUBLICATIONS
Young RM, Hawkins TA, Cavodeassi F, Stickney HL, Schwarz Q, Lawrence LM, Wierzbicki C, Cheng BY, Luo J, Ambrosio EM, Klosner A, Sealy IM, Rowell J, Trivedi CA, Bianco IH, Allende ML, Busch-Nentwich EM, Gestri G, Wilson SW.
Compensatory growth renders Tcf7l1a dispensable for eye formation despite its requirement in eye
field specification.
Elife. 2019 Feb 19;8. pii: e40093. doi: 10.7554/eLife.40093.
Leonardo E. Valdivia, Dayna B. Lamb, Wilson Horner, Claudia Wierzbicki, Amanuel Tafessu, Audrey M. Williams, Gaia Gestri, Anna M. Krasnow, Terra S. Vleeshouwer-Neumann, McKenzie Givens, Rodrigo M. Young, Lisa M. Lawrence, Heather L. Stickney, Thomas A. Hawkins, Quenten P. Schwarz, Florencia Cavodeassi, Stephen W. Wilson, Kara L. Cerveny (2016)
Antagonism between Gdf6a and retinoic acid pathways controls timing of retinal neurogenesis and growth of the eye in zebrafish.
Development 2016 143: 1087-1098; doi: 10.1242/dev.130922
Bazin-Lopez N, Valdivia LE, Wilson SW, Gestri G (2015)
Watching eyes take shape.
Curr Opin Genet Dev. 2015 Mar 3;32C:73-79. doi: 10.1016/j.gde.2015.02.004.
Hernández-Bejarano M, Gestri G, Spawls L, Nieto-López F, Picker A, Tada M, Brand M, Bovolenta P, Wilson SW, Cavodeassi F. (2015)
Opposing Shh and Fgf signals initiate nasotemporal patterning of the zebrafish retina.
Development. 2015 Nov 15;142(22):3933-42. doi: 10.1242/dev.125120.
Miesfeld JB, Gestri G, Clark BS, Flinn MA, Poole RJ, Bader JR, Besharse JC, Wilson SW, Link BA. (2015)
Yap and Taz regulate retinal pigment epithelial cell fate.
Development. 2015 Sep 1;142(17):3021-32. doi: 10.1242/dev.119008.
Cavodeassi F, Ivanovitch K, Wilson SW. (2013)
Eph/Ephrin signalling maintains eye field segregation from adjacent neural plate territories during forebrain morphogenesis.
Development. 2013 Oct;140(20):4193-202.
Ivanovitch K, Cavodeassi F, Wilson SW (2013)
Precocious Acquisition of Neuroepithelial Character in the Eye Field Underlies the Onset of Eye Morphogenesis.
Developmental Cell, October 2013
Gestri G, Link BA, Neuhauss SC. (2012)
The visual system of zebrafish and its use to model human ocular diseases.
Dev Neurobiol. 2012 Mar;72(3):302-27.
K. Cerveny; M. Varga; SW Wilson. (2012)
Continued growth and circuit building in the anamniote visual system.
Developmental Neurobiology 72-3: 328-345
Lupo G, Gestri G, O'Brien M, Denton RM, Chandraratna RA, Ley SV, Harris WA, Wilson SW. (2011) Retinoic acid receptor signaling regulates choroid fissure closure through independent mechanisms in the ventral optic cup and periocular mesenchyme.
Proc Natl Acad Sci U S A. 108(21):8698-703. Epub 2011 May 9.
Cerveny, K.L., Cavodeassi, F., K. J. Turner, T. A. de Jong-Curtain, J. K. Heath, and S. W. Wilson (2010) The zebrafish flotte lotte mutant reveals that the local retinal environment promotes the differentiation of proliferating precursors emerging from their stem cell niche.
Development 137: 2107-2115
McMahon (*), C., Gestri, G. (*), Wilson, S.W. and Link, B.A. (2009)
Lmx1b is essential for survival of periocular mesenchymal cells and influences Fgf-mediated retinal patterning in zebrafish.
Dev Biol. 332(2):287-298.
Gestri G, Osborne RJ, Wyatt AW, Gerrelli D, Gribble S, Stewart H, Fryer A, Bunyan DJ, Prescott K, Collin JR, Fitzgerald T, Robinson D, Carter NP, Wilson SW, Ragge NK. (2009)
Reduced TFAP2A function causes variable optic fissure closure and retinal defects and sensitizes eye development to mutations in other morphogenetic regulators.
Hum Genet. 126:791-803.
Picker A, Cavodeassi F, Machate A, Bernauer S, Hans S, Abe, G., Kawakami, K., Wilson, S.W. and Brand, M. (2009)
Dynamic coupling of pattern formation and morphogenesis in the developing vertebrate retina.
PLoS Biol 7(10)
Gestri,G., Carl,M., Appolloni,I., Wilson,S.W., Barsacchi,G., Andreazzoli,M. (2005)
Six3 functions in anterior neural plate specification by promoting cell proliferation and inhibiting Bmp4 expression.
Development 132:2401-2413
Cavodeassi, F., Carreira-Barbosa, F., Young, R.M., Concha, M.L., Allende, M.L., Houart, C., Tada, M., Wilson, S.W. (2005)
Early Stages of Zebrafish Eye Formation Require the Coordinated Activity of Wnt11, Fz5, and the Wnt/ -Catenin Pathway.
Neuron 47:43-56
Masai,I.,Lele,Z., Yamaguchi,M.,Komori,A.,Nakata,A.,Nishiwaki,Y.,Wada,H.,Tanaka,H., Nojima,Y.,Hammerschmidt,M., Wilson,S.W.,Okamoto,H. (2003)
N-cadherin mediates retinal lamination, maintenance of forebrain compartments and patterning of retinal neurites.
Development 130:2479-2494
Take-Uchi,M.,Clarke,J.D.W.,Wilson,S.W. (2003)
Hedgehog signaling maintains the optic stalk/retinal interface through the regulation of Vax gene activity.
Development 130:955-968
Masai,I.,Stemple,D.L.,Okamoto,H.,Wilson,S.W. (2000)
Midline signals regulate retinal neurogenesis in zebrafish.
Neuron 27:251-263