Myriam Roussigne and Steve Wilson
Nodal signalling imposes left-right asymmetry upon neurogenesis in the habenular nuclei
The left and right sides of our brain mediate different cognitive and sensory functions; for instance, the left hemisphere is dominant in processing aspects of language and tool use while the right hemisphere handles visuospatial attention and interpretation of emotion. This feature of the brain is known as functional lateralisation and is thought to improve our cognitive performance as the specialization of each hemisphere broadens the repertoire of tasks that can be undertaken. Brain lateralisation is not unique to humans and underlies asymmetric behaviour in all vertebrates, a common example being visual lateralization, which is manifest as an eye preference that is dependent on what is being looked at.
Functional lateralisation of the brain is a consequence of structural differences between the left and right sides of particular brain regions. One of the best characterised neuroanatomical asymmetries is found in a region of the brain called the epithalamus and in the last ten years, studies of developing zebrafish have led the way in elucidating the mechanisms by which these epithalamic asymmetries develop. The zebrafish epithalamus consists of several groups of neuron (neuronal nuclei) - the pineal complex and a pair of habenular nuclei which display left-right (LR) asymmetries in the proportion of different subtypes of neurons and in their connections (see the publication summary entitled "Brain asymmetry at the level of single cells"). The pineal complex is composed of the symmetrically positioned epiphysis and a left-sided parapineal nucleus that migrates from the midline and connects to the left habenula. The left-sided migration of the parapineal nucleus is dependent upon the activity of a protein called Fgf8 expressed bilaterally in the presumptive habenulae (see the publication summary entitled "Fgf8 signalling breaks symmetry in the brain"). As it migrates, the parapineal sends signals to the left habenula promoting "left-sided" development by these neurons. If the parapineal is removed, the asymmetry between the left and right habenula is considerably reduced.
The sidedness/laterality of parapineal migration and habenulae asymmetries are always concordant (both on the same side) and dependent on the earlier left-sided signalling by Nodal proteins. Importantly, in the absence of one-sided Nodal signalling, neuroanatomical asymmetries develop but with a randomised sidedness: the parapineal migrates to the left or to right side of the epithalamus with an equal probability and the pattern of habenular asymmetry is either normal or mirror reversed correlating with parapineal position. Thus, Nodal signalling is not required for the establishment of structural asymmetry per se but is responsible of the directionality/laterality of asymmetries.
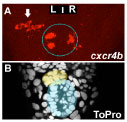
Figure 1: Temporal LR asymmetry in habenular neurogenesis. Images looking down onto the midline of a zebrafish embryo brain. A : cxcr4b expressing neurons appear earlier in the left habenula (arrow) than in the right. The epiphysis is indicated by a dotted blue circle. A’ Staining of all cells in the same brain allows detection of the parapineal (yellow shading) and pineal (blue shading).
The manner in which Nodal signaling imposes left bias in the brain is not known. To address this issue, we explored the involvement of Nodal signalling in promoting the production (neurogenesis) of habenular neurons. We found that a novel marker of habenular precursors/neurons called cxcr4b appears earlier in the left habenula than in the right. This is consistent with previous results from our colleague Hitoshi Okamoto's team showing temporal LR asymmetries in the generation of neuronal sub-types between left and right habenulae. Asymmetry in habenular neurogenesis can be detected before the migration of the parapineal and, indeed, we found that it is independent of the parapineal. In contrast, when we remove the L/R bias in Nodal signaling, habenular neurons appear at the same time in both habenular nuclei. Thus, the activity of Nodal signals in the left epithalamus can directly drive an asymmetry in habenular neurogenesis and this is the first example for a role for Nodal signalling in promoting an asymmetry per se rather than in directing laterality in the brain.
Figure 2. Nodal signalling promotes early asymmetric neurogenesis Images looking down onto the midline of embryonic zebrafish brains showing neurons in green. A and C show normal brains with LR asymmetric neurons (arrows). B and D show brains that don’t have asymmetric Nodal signalling and have LR symmetric neurons (small arrows). The epiphysis is indicated by a dotted blue circle.
The roles/consequences of this Nodal dependent LR asymmetry in habenular neurogenesis are not clear yet but it could be a way by which Nodal biases the orientation of parapineal migration and subsequent habenular asymmetries. This model is consistent with previous data suggesting that the left habenula may provide cues that influence the orientation of parapineal migration. This early asymmetric neurogenesis could also partly influence the later identity of habenular neurons and could consequently be responsible for subtle differences that remain between the left and right habenulae after removal of the parapineal nucleus (see the publication summary entitled "Brain asymmetry at the level of single cells").
How might an early neurogenesis in the left habenula influence the orientation of migration of the parapineal? As we have shown that the protein Fgf8 is required for parapineal migration (see the publication summary entitled "Fgf8 signalling breaks symmetry in the brain"), it will be important to determine if Nodal signalling biases levels of Fgf8 activity, for instance by promoting a L/R asymmetry in the number of fgf8 expressing habenular cells. An alternative possibility is that both asymmetric neurogenesis and directed parapineal migration might be independent consequences of a Nodal-dependent asymmetry in Fgf activity. Our ongoing research is addressing the interactions between Nodal and Fgf signalling pathways in the epithamus.
If you have any further questions, please contact Myriam Roussigne or Steve Wilson
This study was a collaboration with Patrick Blader and our work received financial support from the Wellcome Trust and FEBS.