Forebrain > Diencephalon > Epithalamus> Pineal Complex
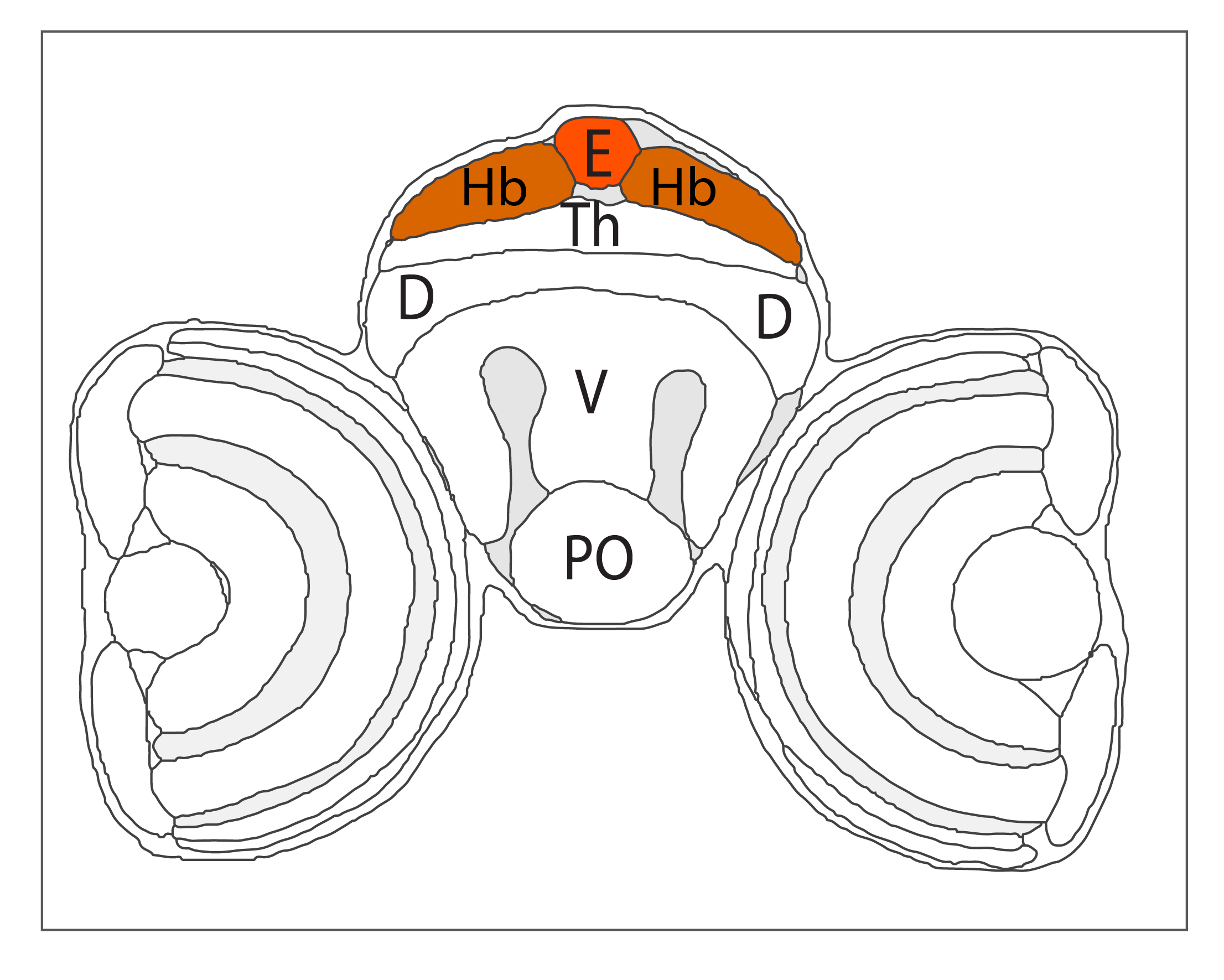
Schematic showing the position of the pineal/epiphysis in sagittal, horizontal and coronal sections through the zebrafish brain.
Based on the anatomical segmentation of 3 day old zebrafish larval brain by Thomas Müller, Olaf Ronneberger, Wolfgang Driever and colleagues. For details see Ronneberger et al., Nat. Meth. 2012 and http://vibez.informatik.uni-freiburg.de
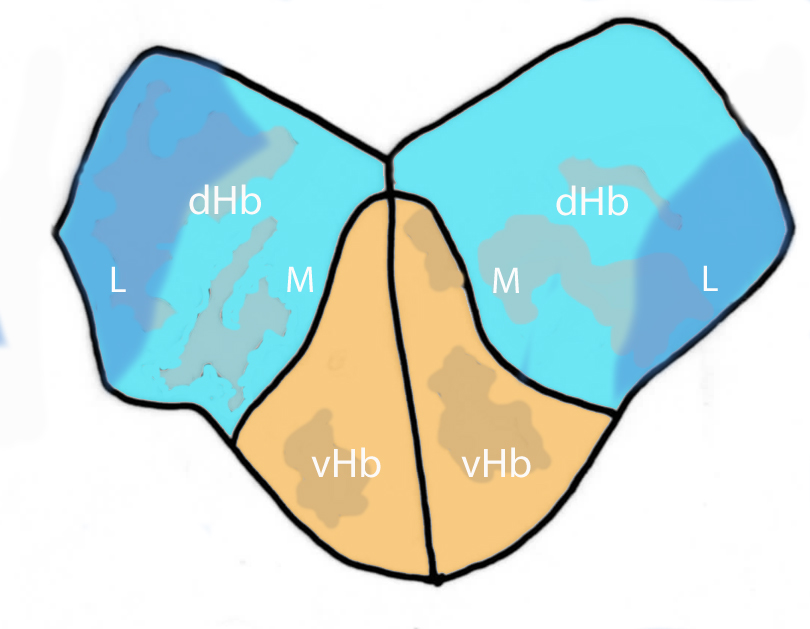
Abbreviations: Ce, cerebellar plate; D, dorsal telencephalon/pallium; E, epiphysis; EmT, eminentia thalami; Hb, habenula; Hyp, hypothalamus; MO, medulla oblongata; OB, olfactory bulb; OT, optic tectum; PG, preglomerular complex; PO, preoptic area;PrT, pretectum; PTd, posterior tuberculum dorsal part; PTh, prethalamus; PTv posterior tuberculum ventral part; Teg, tegmentum; Th, thalamus; TS, torus semicircularis; V, ventral telencephalon/subpallium; Va, valvula cerebelli.
Description
ZFIN
OLS Tree Diagram
Anatomical segmentation of 3 day old zebrafish larval brain by Thomas Müller, Olaf Ronneberger, Wolfgang Driever and colleagues. For details see Ronneberger et al., Nat. Meth. 2012 and http://vibez.informatik.uni-freiburg.de
Transgenic Lines that label this brain region
Antibodies that label this brain region
Key Publications
Back to Forebrain